 
For example, HF, CaF 2, AlF 3Ĩ) In most compounds, the oxidation of oxygen is -2. 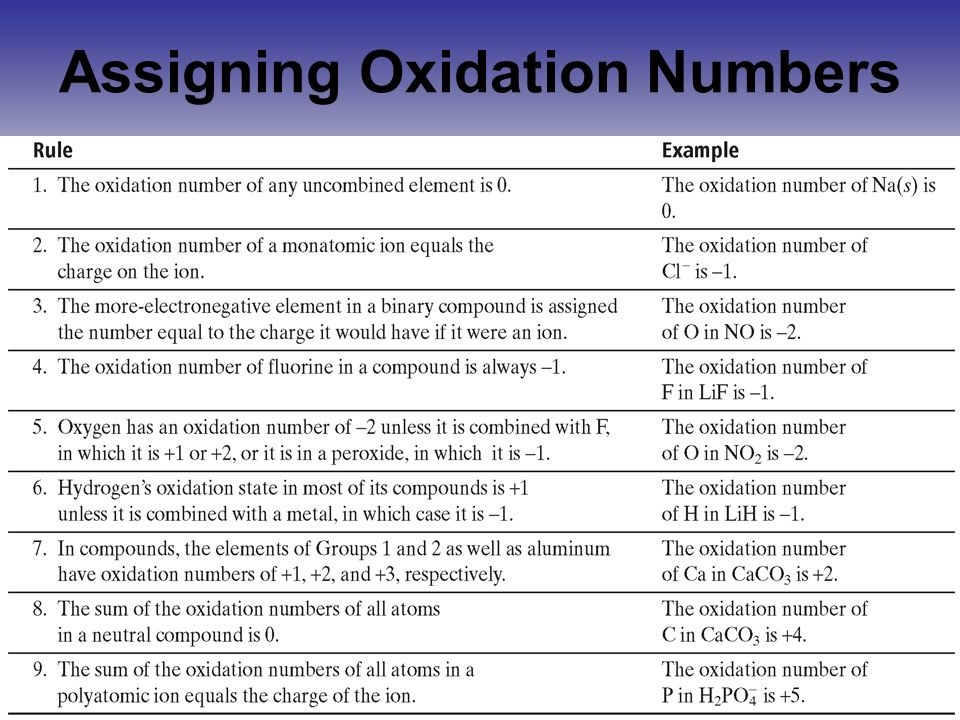
See rule 3.ħ) In its compounds, fluorine is always assigned -1 as its oxidation number. 
Group 1A metals: +1, group 2A metals: +2.Ħ) In compounds with nonmetals, hydrogen is assigned +1 as its oxidation number. The oxidation state is not an actual charge, but rather an assigned, descriptive number.ġ) The oxidation state of an atom in a free element is 0.Ģ) The oxidation state of a monoatomic ion is equal to its charge.ģ) The sum of the oxidation states in a neutral molecule is 0.Ĥ) The sum of the oxidation states in an ion is equal to the charge of the ion.ĥ) Metals have positive oxidation states.Hydrogen has a lower affinity for electrons, and it gives the electron to oxygen, thus the oxidation number of hydrogen in +1. Therefore, the oxidation number of oxygen is -2. Oxygen has a higher affinity for electrons, so it pulls one electron from each bond. Each bond with a hydrogen brings the oxygen -1 oxidation state, so two O-H bonds make its oxidation state -2. Remember, oxygen is more electronegative than hydrogen, and therefore, it is electron-reach while the hydrogens are electron-poor. The general principle is that the more electronegative atom pulls the electron density of the bond toward it and thus it is electron-rich while the other atom is electron-poor.įor example, in water the oxygen is connected to two hydrogen atoms. The oxidation state (or oxidation number) is assigned to each atom in a compound as an indicator of whether the atom is neutral, electron-rich, or electron-poor.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
